September 2024 Case Study
Authors: Nidhi Shah, MD1 and Karl Saardi, MD1
- Department of Dermatology, George Washington University School of Medicine and Health Sciences
Patient History
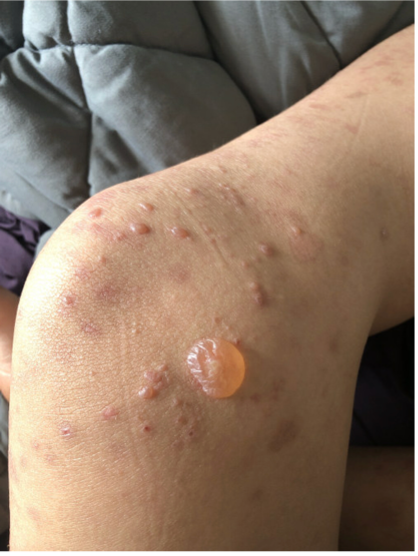
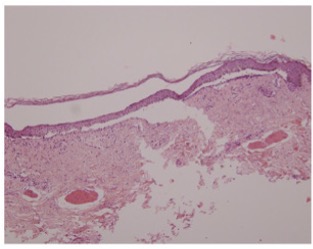
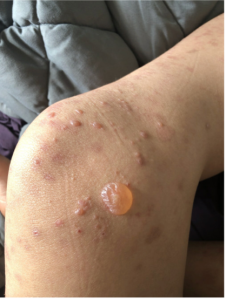
A 27-year-old male with no past medical history presented with a 1-year history of recurrent blisters throughout his body. Figure 1 demonstrates physical examination findings; there was no involvement of the oral or ocular mucosa. A punch biopsy for H&E was performed (Figure 2), and direct immunofluorescence (DIF) on salt-split skin demonstrated dermal staining.
Based on the clinical presentation and biopsy findings, what is the target for the circulating and tissue-bound autoantibodies?
A. LAD-1
B. Collagen VII
C. Laminin 332
D. BPAG2
E. Transglutaminase
Correct Answer: B
Explanation/Literature Review
Given the formation of tense bullae and vesicles in a young adult with biopsy findings demonstrating subepidermal cleavage with sparse inflammatory infiltrate and salt-split skin revealing staining on the dermal side, the patient was diagnosed with Epidermolysis Bullosa Acquisita (EBA), a very rare autoimmune blistering condition. The condition can be acquired at any age, with the median age of 50 years.1 EBA is characterized by development of autoantibodies against collagen VII, which is a major component of anchoring fibrils in the hemidesmosomes found in the sub-lamina densa of the skin and mucous membranes.1,2 On histology, there is subepidermal cleavage with or without inflammatory infiltrate based on the subtype of EBA. Further immunofluorescence studies demonstrate linear deposition of IgG and C3 at the basement membrane zone, with a u-serrated pattern and fluorescence only on the dermal side.1,2 Salt split skin DIF is a challenging technique requiring an experienced immunodermatology lab, in which patient’s skin is preserved in IF media, treated with IM NaCl, and then processed for DIF. This technique can be especially helpful in diseases where IIF is negative, and ELISA assays are not available.3 Traditionally, immunoelectron microscopy is considered the “gold standard” for diagnosing EBA.
LAD-1 (choice A) and LABD97 are the targets for linear IgA bullous dermatosis (LABD) which generally affects elderly adults and preschool-aged children and has histopathology findings of IgA +/- C3 deposition along basement membrane with epidermal staining on salt-split skin.4 Laminin 332 (choice C) is the target for anti-epiligrin mucous membrane pemphigoid (MMP) which on salt-split skin also demonstrates dermal deposition of IgG, similar to EBA, however, the condition is classically seen in elderly patients and the lack of mucosal involvement makes the diagnosis less likely.5,6 In bullous pemphigoid the targets include BPAG1 and BPAG2 (choice D), and clinically the presentation can be similar to the inflammatory subtype of EBA, however, on histology there would linear C3 and IgG deposition along the basement membrane in an “n-serrated” pattern with mostly epidermal staining on salt-split skin.7 Epidermal transglutaminase (choice E) is the target for dermatitis herpetiformis (DH), an autoimmune disease which always occurs in conjunction with celiac disease. IIF in DH is negative, and on DIF DH is differentiated from LABD by granular vs. linear IgA deposition at the basement membrane.8
Given the rarity of the condition and lack of randomized controlled trials, treatment is challenging and is based on case reports and findings from other autoimmune blistering conditions. First line options include systemic corticosteroids, IVIG, and immunomodulators including colchicine and dapsone, with immunosuppressants such as mycophenolate mofetil, azathioprine, and cyclosporine reserved as second line.1,9 Often, EBA is resistant to these therapies and recent evidence supports the use of rituximab.1,9
References
- Kridin K, Kneiber D, Kowalski EH, Valdebran M, Amber KT. Epidermolysis bullosa acquisita: A comprehensive review. Autoimmun Rev. 2019;18(8):786-795. doi:10.1016/j.autrev.2019.06.007
- Caux F. Diagnosis and Clinical Features of Epidermolysis Bullosa Acquisita. Dermatol Clin. 2011;29(3):485-491. doi:10.1016/j.det.2011.03.017
- Bolognia J, Schaffer JV, Cerroni L, eds. Dermatology. Fifth edition. Elsevier; 2025.
- Hull C, Zone J. Dermatology. In: Bolognia J SJCL, ed. Dermatology. Vol 31. e1 ed.; 2018:457-537.
- Shi L, Li X, Qian H. Anti-Laminin 332-Type Mucous Membrane Pemphigoid. Biomolecules. 2022;12(10):1461. doi:10.3390/biom12101461
- Carey B, Setterfield J. Mucous membrane pemphigoid and oral blistering diseases. Clin Exp Dermatol. 2019;44(7):732-739. doi:10.1111/ced.13996
- Miyamoto D, Santi CG, Aoki V, Maruta CW. Bullous pemphigoid. An Bras Dermatol. 2019;94(2):133-146. doi:10.1590/abd1806-4841.20199007
- Witte M, Zillikens D, Schmidt E. Diagnosis of Autoimmune Blistering Diseases. Front Med (Lausanne). 2018;5:296. Published 2018 Nov 2. doi:10.3389/fmed.2018.00296
- Miyamoto D, Gordilho JO, Santi CG, Porro AM. Epidermolysis bullosa acquisita. An Bras Dermatol. 2022;97(4):409-423. doi:10.1016/j.abd.2021.09.010